POTASSIUM CHLORIDE
EXTENDED-RELEASE TABLETS, USP
PRODUCT DETAILS
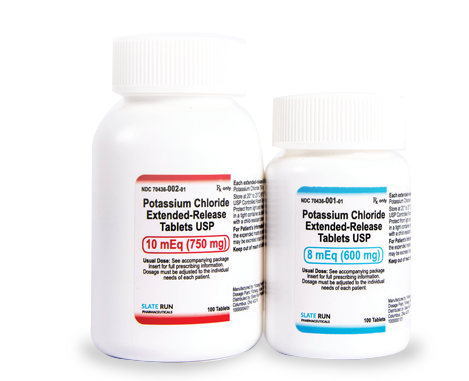
- Strength: Available in 600 mg (8 mEq) and 750 mg (10 mEq) tablets
- Therapeutic Class: Electrolyte Supplement
NDC Numbers:
600 mg | Bottle of 100: 70436-001-01
600 mg | Bottle of 1000: 70436-001-03
750 mg | Bottle of 100: 70436-002-01
750 mg | Bottle of 1000: 70436-002-03
Available through your wholesaler:
NDC: 70436-001-01
Cardinal Health: 5520010
McKesson: 3905932
Amerisource: 10213954
NDC: 70436-001-03
Cardinal Health: 5549191
McKesson: 3905999
Amerisource: 10214188
NDC: 70436-002-01
Cardinal Health: 5549209
McKesson: 3906005
Amerisource: 10213953
NDC: 70436-002-03
Cardinal Health: 5514658
McKesson: 3906013
Amerisource: 10222983
| Strength | NDC | Package Size |
|---|---|---|
| 600 mg (8 mEq) | 70436-001-01 | Bottle of 100 |
| 600 mg (8 mEq) | 70436-001-03 | Bottle of 1000 |
| 750 mg (10 mEq) | 70436-002-01 | Bottle of 100 |
| 750 mg (10 mEq) | 70436-002-03 | Bottle of 1000 |
Potassium Chloride Extended-Release Tablets USP are supplied as round, white to off-white tablets. The 600 mg is debossed with “YH” and “106” on one side, and “8” on the other side. The 750 mg is debossed with “YH” and “105” on one side, and “10” on the other side.
Store at 20° to 25°C (68° to 77°F). [see USP Controlled Room Temperature]. Protect from light and moisture. Dispense in a tight container as defined in the USP with a child-resistant closure.
